Selectivity Factor Calculator
Introduction
Calculators are essential tools for various mathematical computations. This article presents a JavaScript calculator designed to calculate the selectivity factor for a given set of data. With its user-friendly interface and accurate formula, this calculator simplifies the process of determining selectivity factor values.
How to Use
To utilize the calculator, input the required values into the designated fields and click the “Calculate” button. The calculator will then compute the selectivity factor based on the provided data.
Formula
The selectivity factor (SF) is calculated using the following formula:
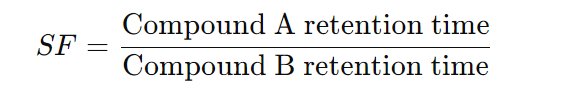
Where:
- SF is the selectivity factor.
- Compound A retention time is the time taken for Compound A to pass through a chromatographic column.
- Compound B retention time is the time taken for Compound B to pass through the same chromatographic column.
Example Solve
Suppose Compound A has a retention time of 10 seconds, and Compound B has a retention time of 5 seconds. To find the selectivity factor:
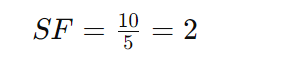
Thus, the selectivity factor is 2.
FAQs
Q: What is the significance of the selectivity factor in chromatography?
A: The selectivity factor helps determine the separation efficiency between two compounds in chromatography. It indicates the degree of separation achieved by the chromatographic method.
Q: How is the selectivity factor used in analytical chemistry?
A: Analytical chemists use the selectivity factor to assess the selectivity of a chromatographic system and optimize separation conditions for accurate compound analysis.
Q: Can the selectivity factor be greater than 1?
A: Yes, a selectivity factor greater than 1 indicates good separation between compounds, while a value closer to 1 suggests minimal separation.
Conclusion
The selectivity factor calculator presented here offers a straightforward solution for determining the selectivity factor in chromatography. By inputting the appropriate retention times, users can quickly obtain accurate selectivity factor values, aiding in the optimization of chromatographic separations.
